Graphene Biosensors
A biosensor is a device which measures biological or chemical reactions by creating an electrical output response that is proportional to the concentration of a particular substance (or analyte) within that reaction. They are used in a wide range of areas including medical treatment, environmental surveys, food control, forensics and research.
What Makes an Ideal Biosensor?
For every single type of biosensor, an optimally performing device will feature five characteristics, able to:
- Detect a specific substance in a sample mixed with other chemical compounds
- Produce identical responses within duplicated experiments
- Resist unwanted effects from internal and external disturbances such as temperature changes or degradation of the bioreceptor
- Detect substances in concentrations as low as on the ng/ml (or even fg/ml) scales
- Produce a straight line relationship between concentration and output response to a high resolution and over a wide enough range

Other physical attributes necessary if a biosensor is to be used in the real world space are that it has to be portable and user friendly. Additionally, if it comes with a probe for invasive monitoring by doctors, nurses or other medical professionals, the probe must be thin and biocompatible.
Graphene & Biosensors: A Perfect Match
Graphene has emerged as the new “wonder material” for biosensors thanks to its unique physical and chemical properties. It has an electrical conductivity of the order of 1,000 siemens per meter and thermal conductivities between 1,500 and 2,500 Wm−1K−1. It is the strongest material ever tested, too, with a tensile strength around 130 GPa. As a comparison, silicon has an electrical conductivity of 1,000 siemens per metre, a thermal conductivity of 150 Wm-1K-1, and a tensile strength of around 170 MPa.
Graphene exhibits a broad electrochemical window of approximately 2.5 V in 0.1 moles per litre of phosphate buffered saline. It also has a low charge-transfer resistance around 6.5 MΩ cm2, meaning it is an ideal material for use in multifunctional fast sensors. Graphene is able to be functionalized through the addition of functional groups which affect its reactivity. If bioreceptors are added, this makes graphene ideal for use in biosensors. It is also biocompatible, perfect for clinical or medical uses, and is cheap, safe and relatively easy to fabricate.
Since graphene was only discovered in 2004, more research is needed to create fabrication methods that are even better than they are now. The underlying science behind graphene is still not fully understood as well, with further work required in areas such as the absorption mechanism of molecules on graphene surfaces, the orientation of biomolecules on the graphene and how these interactions affect graphene’s transport properties.
What Are GFETs and SPPs?
The favorable qualities of graphene have led to a wide array of investigations into its use in biosensing. Two particularly interesting configurations are graphene field-effect transistors (GFETs) and graphene enhanced surface plasmon polaritons (SPPs).
GFETs
GFETs are a modification of the classic silicon field effect transistor. In traditional transistors, silicon acts as a thin conducting channel, the conductivity of which can be tuned with applied voltage. GFETs perform in a similar manner, with the silicon replaced with graphene. This yields a much thinner, more sensitive channel region. Due to graphene’s broad electrochemical potential and its ability to be functionalized, GFETs present an attractive device for biomolecules to attach to. Because of graphene's extreme surface-to-volume ratio, even the smallest concentration of attached molecules changes the channel conductivity. GFET biosensors are a good platform for sensing a wide variety of species such as enzymes, hydrogen peroxides, dopamine, and reduced b-nicotinamide adenine dinucleotide (NADH) molecules.
SPPs
Graphene can also be used in conjunction with surface plasmon polaritons (SPPs) on metal films to enhance biosensor performance. SPP-based sensors utilize the confinement of optical waves on the surface of metals to make small-volume chemical and biological sensors. The sensing volume is given by the tightly confined surface wave, boosting sensitivity of optical detection. Standard metals used in SPPs are gold (Au) and silver (Ag) due to their favorable propagation properties. However, silver corrodes quickly, and gold has poor adsorption properties. Layering graphene on top of the gold, however, results in superior adsorption.
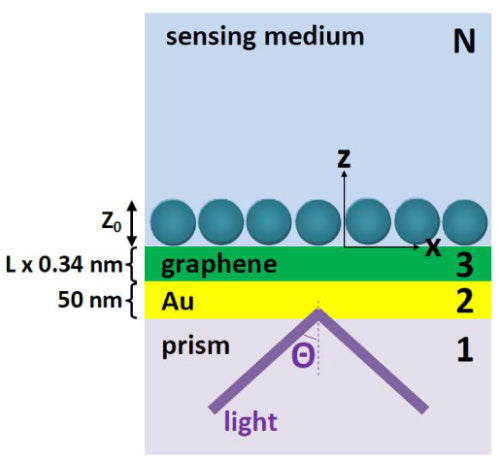
Figure: SPP sensor enhanced with graphene (Reproduced from Optics Express 18, 14395 (2010) with permission from the Optical Society of America).
Graphene: Expanded Horizons
Due to its versatility, graphene can be used in a range of sensors beyond GFETs and SPPs. In fact, a large number of other graphene-enhanced sensors are on the rise, including microelectromechanical systems (MEMS) sensors, pressure sensors, pH sensors, environmental contamination sensors, gas sensors, DNA sensors and more.
Certain researchers are thinking more creatively, too, around how to use graphene in sensors. Materials science research center AMBER at Trinity College Dublin has experimented with adding graphene to silly putty with the resulting nanocomposites displaying unusual behavior. The resulting device was a highly sensitive sensor with gauge factors of greater than 500 which could measure everything from blood pressure and pulse to the impact of a spider’s footsteps.
Graphenea Offer A Range of Graphene-Based Field Effect Transistors in different configurations for research purposes. See the full range here, or contact us today to discuss any specific configurations you need.

